Innovating Inside the Box: Adapting a Platform Drug Delivery Device

We’re always told to “think outside the box.” But when it comes to drug delivery devices, sometimes the real challenge is innovating inside the box—that is, working within constraints to adapt off-the-shelf devices to unique requirements.
Drug Delivery Decisions: Adopt, Create or Adapt
Drug developers have three options when it comes to matching a drug with a delivery device:
-
Adopt an off-the-shelf drug delivery device (use an existing box)
-
Create a completely custom device (build a new box)
-
Adapt an existing platform device with custom attributes (innovate inside the box)
Adapting an existing platform device is cheaper and faster than creating a custom device from scratch. Often, small adaptations can be made to address specific user requirements, drug formulation challenges or business priorities.
But the choice between adopting, creating and adapting isn’t always clear. Device engineers must carefully consider the drivers for customization—including the drug formulation and administration routes, special user needs, and market and pipeline considerations. Then, they must weigh the pros and cons of each development option.
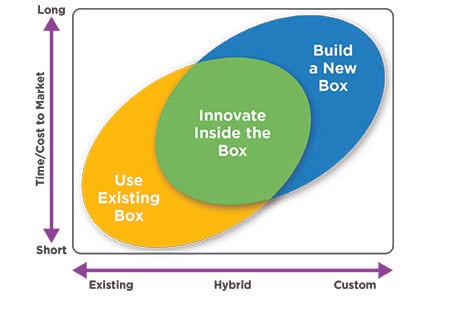
How to Innovate Inside the Box
“Innovating inside the box” has its own challenges. Each off-the-shelf drug delivery device has its own constraints and design limitations. When making a change to the device, engineers must evaluate the positive and negative impacts, including newly introduced risks and regulatory implications.
Battelle helps drug manufacturers make informed decisions about drug delivery device development and minimize risks, costs and timelines for adapting off-the-shelf devices.
You can learn more about our approach to drug delivery device innovation in our new whitepaper. You’ll learn:
-
How to weigh the pros and cons of adopting, creating or adapting a medical device
-
The drivers for device customization
-
How to minimize or mitigate introduced risks when adapting an off-the-shelf device
-
What to look for in a development partner for drug delivery device adaptation

Related Blogs
BATTELLE UPDATES
Receive updates from Battelle for an all-access pass to the incredible work of Battelle researchers.