New Approach Methods: The Future of Biomedical Research
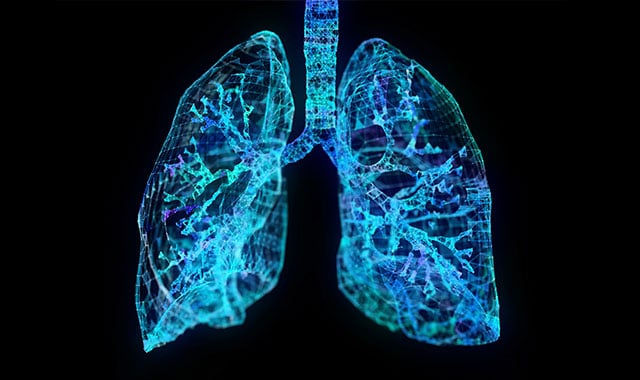
In recent years, regulatory agencies, including the EPA and FDA have begun to consider new approach methods (NAMs) and modeling technologies. These new tactics aim to reduce the time and expense associated with toxicology testing while also finding more ethical ways to conduct clinical trials.
A New Approach to Testing
This shift has sparked a growing interest in developing new ways to replace traditional toxicology testing. Increases in computing speed have made the use of simulation and modeling technologies for regulatory approval more widely available and cost-effective.
One frontrunner is in silico modeling, which uses computer models to simulate biological processes. This approach saves valuable time and resources, predicting side effects and risks before moving on to the next stage of clinical trials. Simulations run on the order of a day or less, whereas traditional testing can take months and years to complete.
Other approaches include 3-D cell cultures or organoids. Organoids provide a more accurate representation of human biology when studying drug efficacy and toxicity. Microfluidic devices, or "organ-on-a-chip", contain small channels that allow researchers to mimic human tissue.
As modeling technologies advance, researchers can increase the sample size for statistical significance without testing on living organisms.

The Future of Health Research
New approaches in toxicology testing are shaping the future of health by enabling accurate, reliable, and cost-effective research.
Cost and time effective
Conventional methods can take years and can cost millions of dollars. New Approach Methods (NAMs), which include computational modeling and in vitro assays are a fraction of the cost and can be run simultaneously. This means that multiple conditions can be assessed in parallel by running numerous models at the same time, to deliver faster results.
NAMs refer to technologies and methodologies or combination approaches used to perform hazard and risk assessment. For researchers, this means greater potential to accelerate the regulatory process by eliminating failures earlier in the development process.
Increased Accuracy
Since NAMs are based on human biology, they provide a more accurate predictor of toxicity, ensuring that drugs are safe and effective in humans.
Personalized Medicine
NAMs allow researchers and drug developers to test drugs on specific populations, or even individual patients.
Battelle's Impact
At Battelle, researchers have been using similar models to conduct research for decades.
In a recent study, Battelle, Syngenta Crop Protection, Greek Creek Toxicokinetics Consulting, LLC, and Pacific Northwest National Lab partnered to study the toxicology of Chlorothalonil. A fungicide used in agriculture since 1966, chemical exposure has been found to cause tissue irritation when present above specific thresholds.
Chlorothalonil was undergoing the pesticide re-registration mandated by the EPA. The program is a safety assessment to identify adverse effects on human health and provide guidance on chemicals used in agriculture.
The team performed computational fluid dynamics (CFD) modeling to simulate exposure to humans and correlate the results to in vivo studies. Previous work has assessed the nasal airways but lacked human oral breathing in the models.
Battelle was able to generate a model of the mouth to the upper airways of the lungs to simulate oral breathing. Breathing was simulated at a rate appropriate for someone working in fields where exposure would likely occur.
This data was combined with a mucociliary clearance model and multiple path particle deposition (MPPD) models to determine an individual's retained dose profile over a five-day work week, at eight hours per day exposure.
The results from this assessment can be applied to model and track different particulate sizes and inform initial assessment of other pesticide compositions and active ingredients.
As expected, smaller particles penetrated deeper into the lungs, while larger particles were deposited in the upper airways. This data was pooled with models of human nasal breathing for a toxicology assessment based on in vitro studies.
This study marks the first time the EPA has accepted the reauthorization of a chemical compound using new approach methodologies. This modeling can be adapted to include medicines such as pressurized metered dose inhalers, dry powder inhalers, and even nasal-administered medications.

Related Blogs
BATTELLE UPDATES
Receive updates from Battelle for an all-access pass to the incredible work of Battelle researchers.