NeuroLife® Sleeve Brings New Approach to Stroke Rehabilitation

Stroke is a leading cause of adult long-term disability1. When the blood supply to the brain is interrupted or reduced, results can be catastrophic, and recovery is extremely limited. Of the 5 million stroke survivors today, 60% are living with a permanent disability due to hand and arm impairment2. And the costs associated with strokes—caregiving, complications and decreased productivity—are estimated at $50 billion per year1.
In the past, options for recovering original function of a limb after stroke were nearly unattainable. Rehabilitation from a stroke can take years, and even then, the results often will provide limited functional results. It is difficult to retrain nerves and muscles to their full potential.
Possibilities With NeuroLife®
Advancing science and knowledge is bringing newfound hope for those living with stroke-related impairments. Nearly a decade ago, when we started work on the Battelle NeuroLife program, we hoped to find an off-the-shelf sleeve that could stimulate muscles in the forearm; the sleeve we wanted would be controlled by a brain-computer interface that read neural signals from a brain implant. But such a sleeve wasn’t on the market, so we invented one of our own for the purpose. Little did we know the potential for this technology.
The Battelle NeuroLife® Sleeve is now in its third generation of form. The electrodes that deliver stimulation to forearm muscles to control movement also read muscle activity to decipher movement intention, instead of a brain implant. We are producing research that shows this technology may help restore independence for people living with stroke-related disabilities by improving lost hand function. We hope that our new technology can provide deeper insights for rehabilitation and those insights are being studied in novel rehabilitation paradigms that were unattainable until we built this technology.
The promise of our research was recognized by a recent award. Our “NeuroLife® Sleeve Development Program–Aiming to Restore Hand Function after Stroke” entry won “Most Innovative” at the American Congress of Rehabilitation Medicine ACRM Annual Conference last month. This work was developed and presented by a Battelle team led by Dr. Eric Meyers and included Dr. Lauren Wengerd, Dr. David Gabrieli, Dr. Michael Darrow, Nick Tacca and Dr. Dave Friedenberg. It is the second year in a row that a Battelle technology has won this award. Last year, our Activity AssistantTM also won “Most Innovative”. 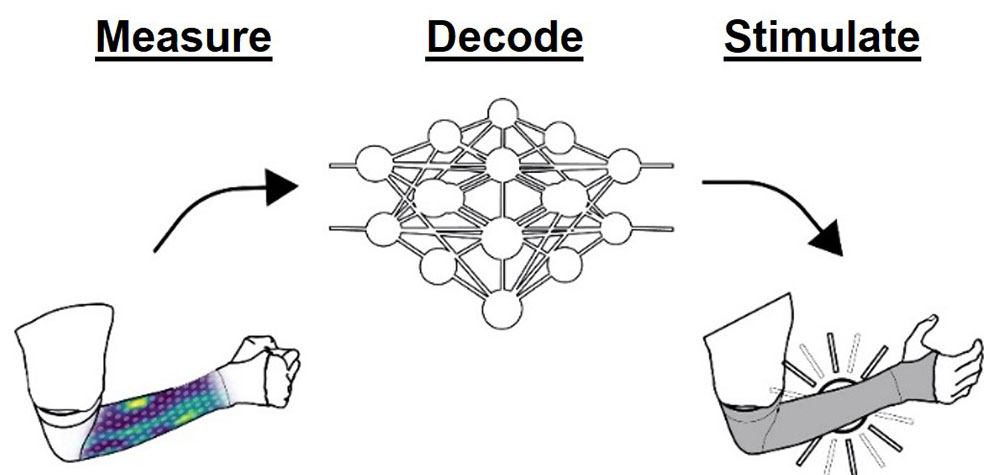
How It Works
When available, the Battelle NeuroLife® Sleeve will be designed to be comfortable and portable. With 150 noninvasive electrodes, it can decode multiple movements and rapidly evoke a variety of hand and forearm movements without having to be connected to a brain-computer interface. We are currently testing if the sleeve can enhance neuroplasticity in those living with stroke-related disabilities to improve long-term recovery.
When worn, the NeuroLife® Sleeve continuously measures muscle activity in the forearm and when a movement intention is detected, the sleeve delivers functional electrical stimulation (FES) to muscles across the forearm, which we believe will enhance a variety of hand and wrist movements.
The user controls the therapy during task-oriented rehabilitation. As we continue to develop this new product, our goal is for daily independence to be attainable and ultimately restore hope for millions of stroke survivors who want their quality of life improved.
What's Next
Battelle will continue its research into this new technology and may require the participation of persons in the Central Ohio region who have different types of disabilities impacting their motor functions. At this time, studies are fully enrolled but future opportunities for Battelle research studies will be shared as they become available.
Disclaimer: NeuroLife® reanimation technology, is an investigational device and is not currently available for sale. It has not been evaluated, cleared, or approved by the U.S. Food and Drug Administration.

Citations
1. Virani, S. S., Alonso, A., Aparicio, H. J., Benjamin, E. J., Bittencourt, M. S., Callaway, C. W., ... & American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. (2021). Heart disease and stroke statistics—2021 update: a report from the American Heart Association. Circulation, 143(8), e254-e743.
2. Langhorne, P., Coupar, F., & Pollock, A. (2009). Motor recovery after stroke: a systematic review. The Lancet Neurology, 8(8), 741-754.
Related Blogs
BATTELLE UPDATES
Receive updates from Battelle for an all-access pass to the incredible work of Battelle researchers.